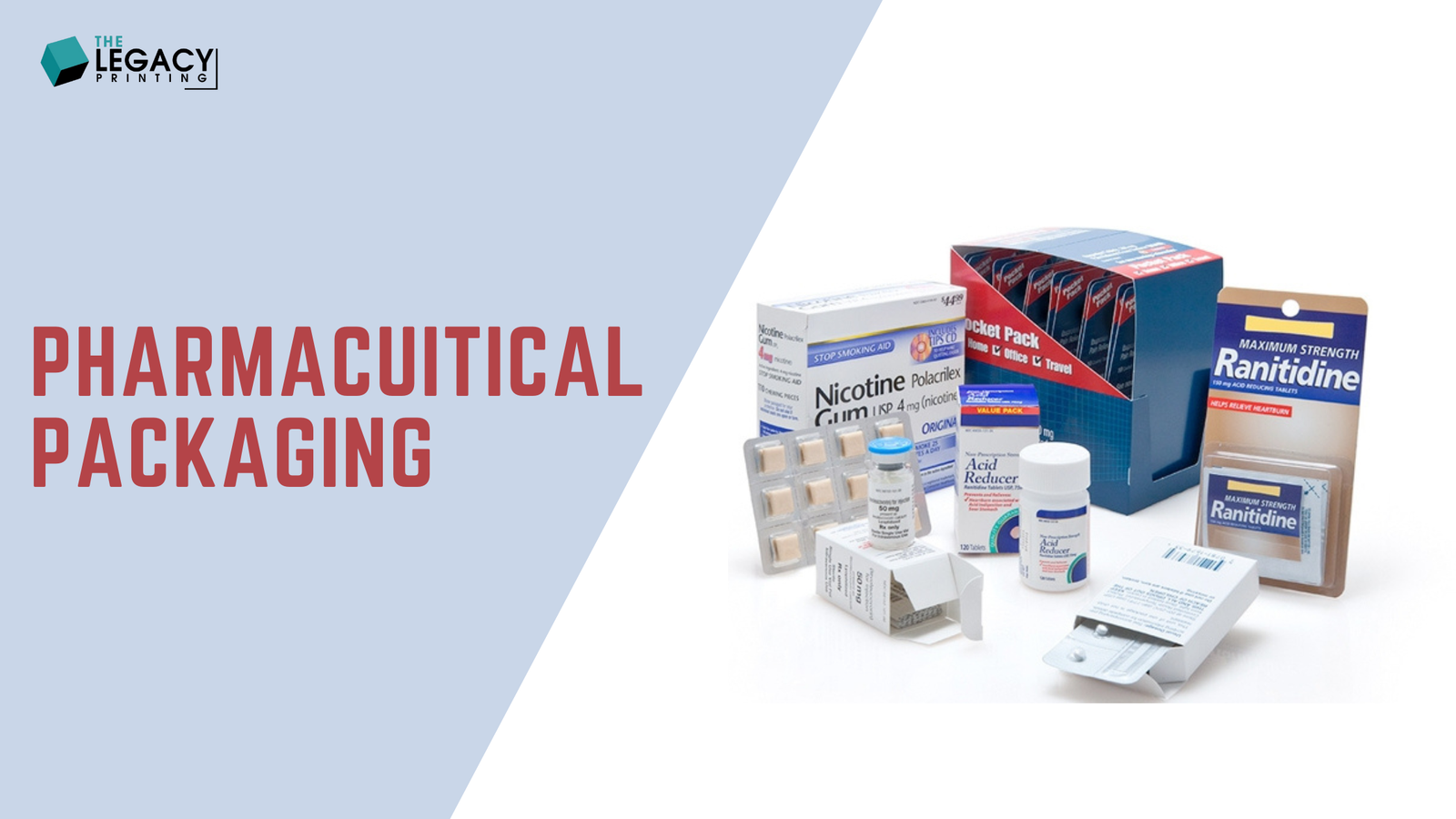
In 2025, pharmaceutical packaging is the backbone of a $1.5 trillion industry, defining how medications are protected, delivered, and experienced by patients across the globe. The global contract packaging market, valued at $17.6 billion in 2024, is projected to grow at a 7.1% CAGR through 2030, fueled by innovation, stringent regulations, and rising consumer expectations. For small businesses, e-commerce pharmacies, and industry professionals, drug packaging is a strategic lever to slash costs, ensure compliance, and differentiate brands in a competitive market. Understanding Drug Packaging
The Core of Pharmaceutical Packaging
Pharmaceutical packaging encompasses the materials and processes used to safeguard, store, and deliver medications, from primary containers like blister packs, vials, and syringes to secondary elements like cartons and labels. Its role extends beyond protection to ensuring product integrity, patient safety, regulatory compliance, and an enhanced user experience. As Justin Schroeder from PCI Pharma Services emphasizes, “Medication packaging must prioritize ease of use, safety, and adherence while maintaining uncompromised quality.” In 2025, drug packaging is not just a necessity but a strategic asset for startups and e-commerce pharmacies looking to scale. The complexity of modern drugs, from biologics to personalized therapies, demands innovative packaging solutions that balance functionality with cost-efficiency, making pharma packaging a critical focus for businesses aiming to thrive.
Why Small Businesses Should Care
For small businesses and e-commerce pharmacies, pharma packaging is a lifeline to competitiveness in a high-stakes industry. Building in-house packaging facilities can cost millions, an insurmountable barrier for startups with limited capital. By partnering with contract packaging organizations (CPOs), small businesses gain access to cutting-edge technologies—such as AI-driven serialization for anti-counterfeiting or eco-friendly materials like post-consumer recycled (PCR) glass—without the hefty upfront investment. Industry data shows that optimized drug packaging can reduce costs by 7-9%, freeing up resources for marketing or product development. For example, a small online pharmacy launching a niche biologic can leverage CPO expertise to ensure compliance with FDA regulations while keeping logistics costs low. Pharmaceutical packaging compliance for small businesses empowers startups to compete with larger players, delivering high-quality, compliant products that build trust and drive growth.
Forces Shaping Pharma Packaging in 2025
The evolution of medication packaging is driven by multiple converging factors. The surge in biologics, biosimilars, and GLP-1 agonists (e.g., weight loss drugs) requires specialized solutions like cold chain pharmaceutical packaging for biologics to maintain drug stability during transport. Regulatory pressures, such as the EU’s Falsified Medicines Directive (FMD), mandate serialization to combat counterfeiting, a $200 billion global issue that threatens patient safety. Meanwhile, consumer demand for sustainability—over 50% of Americans prefer brands with eco-friendly practices—has spurred adoption of recyclable and biodegradable materials. Innovations like SGD Pharma’s 20% PCR glass bottles, introduced in 2024, exemplify this shift. These dynamics position pharmaceutical packaging as a critical strategic focus for businesses aiming to lead in 2025, requiring agility and foresight to navigate a rapidly changing landscape.
Why Pharmaceutical Packaging Matters in 2025
-
Mastering a Complex Industry Landscape
The pharmaceutical industry in 2025 is a labyrinth of challenges and opportunities, with the global packaging market projected to reach $397.71 billion by 2034 at a 10.7% CAGR. Unlike a decade ago, when large corporations dominated, today’s market rewards agility and innovation, thanks to CPOs that democratize access to advanced drug packaging solutions. For small businesses, this shift is a game-changer, enabling them to compete without massive infrastructure investments. By outsourcing pharma packaging, startups can focus on core competencies like product development or customer engagement while leveraging CPO expertise in compliance, sustainability, and technology. This levels the playing field, allowing e-co
mmerce pharmacies to deliver biologics or generics with the same precision as industry giants, making medication packaging a strategic priority for growth.
-
MediQuick’s Transformative Journey
Consider MediQuick, a small e-commerce pharmacy launched in 2023, which faced daunting challenges packaging GLP-1 agonist injections. In-house efforts led to skyrocketing logistics costs and regulatory risks, threatening its survival. By partnering with a CPO specializing in cold chain pharmaceutical packaging for biologics, MediQuick turned the tide. The CPO provided temperature-controlled vials and real-time monitoring, slashing logistics expenses by 15% and ensuring FDA compliance. User-friendly syringe designs boosted customer satisfaction by 25%, driving repeat orders. By 2025, MediQuick scaled to 12,000 monthly orders, a testament to how strategic pharma packaging can transform a startup’s trajectory. This case underscores the power of outsourcing drug packaging to unlock cost savings, compliance, and customer trust.
-
Strategic Advantages for Small Businesses
Optimized medication packaging delivers a trifecta of benefits: cost efficiency, regulatory compliance, and access to cutting-edge innovations like smart pharmaceutical packaging with RFID tags. For small businesses, this means faster market entry, crucial in a fast-paced industry where biologics and generics are booming. CPOs enable startups to adopt advanced solutions—such as IoT-enabled packaging for temperature monitoring or biodegradable blisters—without the R&D burden. By addressing biologics, regulations, and consumer demands for sustainability, pharmaceutical packaging empowers e-commerce pharmacies to build scalable, customer-centric brands. The ability to pivot quickly, whether launching a niche therapy or scaling to meet demand, makes drug packaging an indispensable tool for 2025 success.
Key Trends Shaping Medication Packaging
1-Patient-Centric Packaging: Putting Users First
The Rise of Home-Based Care
In 2025, drug packaging is redefining patient engagement as treatments increasingly shift from clinical settings to homes. This trend is critical for e-commerce pharmacies targeting direct-to-consumer markets, where user experience drives loyalty and retention. Patient-centric pharmaceutical packaging for elderly must be intuitive, safe, and designed to promote adherence, especially for patients managing chronic conditions like diabetes or rheumatoid arthritis. The move toward home-based care, accelerated by telehealth and personalized medicine, demands packaging that empowers patients to manage their therapies independently. For small businesses, investing in patient-centric pharma packaging builds trust and differentiates brands in a crowded market, fostering long-term customer relationships.
Designing with Empathy
For patients with chronic illnesses, medication packaging can make or break adherence. CPOs are pioneering empathetic designs tailored to diverse needs: blister packs with large-print dosing schedules for visually impaired patients, prefilled syringes with ergonomic grips for those with arthritis, and tamper-evident closures that balance child resistance with senior accessibility. In 2024, a startup pharmacy partnered with a CPO to develop blister packs with QR codes linking to video tutorials, boosting adherence by 20% among elderly patients. By 2025, such digital integrations are standard, transforming patient-centric pharmaceutical packaging for elderly into a cornerstone of innovation. These solutions not only improve health outcomes but also enhance brand loyalty, as patients gravitate toward products that feel personalized and accessible.
How Small Businesses Can Implement
To adopt patient-centric drug packaging, small businesses should collaborate with CPOs that employ behavioral psychologists to understand patient pain points. For instance, color-coded blister packs reduce dosing confusion for elderly patients, while tactile indicators aid those with visual impairments. Testing prototypes with real users ensures usability, and integrating digital elements like QR codes or NFC tags enhances engagement by linking to dosage reminders or care plans. For e-commerce pharmacies, this approach drives higher retention and positive reviews, amplifying brand impact. By prioritizing patient needs, small businesses can leverage pharmaceutical packaging to create meaningful connections and stand out in 2025.
2-Sustainability: Leading the Green Revolution
Consumer and Regulatory Pressures
Sustainable pharmaceutical packaging solutions are a defining trend in 2025, with the sustainable packaging market projected to reach $343.01 billion by 2034 at a 15.13% CAGR. Over 50% of consumers prefer brands with eco-friendly practices, while regulations like the EU’s Packaging and Packaging Waste Regulation (PPWR) mandate recyclable materials by 2030. CPOs are responding with innovations like SGD Pharma’s 20% PCR glass bottles, launched in 2024, and biodegradable blister packs made from plant-based plastics. These solutions align with consumer values and regulatory demands, positioning pharma packaging as a leader in the green revolution. Small businesses adopting sustainable drug packaging can attract environmentally conscious customers while future-proofing against stricter regulations.
Balancing Safety with Sustainability
For small businesses, sustainable medication packaging is a double-edged sword: it draws eco-conscious consumers but must meet stringent safety standards to protect drug efficacy. CPOs are tackling this challenge with chemical recycling, producing food-grade recycled plastics for oral medications, and biodegradable films certified under ASTM D6400 for compostability. The UK’s CiPPPA initiative, which recycles drug packaging like blister packs, serves as a global model for circularity, reducing landfill waste. By partnering with CPOs that prioritize both safety and sustainability, small businesses can adopt green solutions without compromising compliance, making sustainable pharmaceutical packaging solutions a strategic advantage in 2025.
GreenRx’s Sustainability Success
GreenRx, a small e-commerce pharmacy specializing in herbal supplements, partnered with a CPO in 2024 to transition to biodegradable blister packs, reducing plastic use by 30%. The move earned GreenRx a “Green Brand” award and boosted sales by 18%, as eco-conscious consumers flocked to their sustainable offerings. By choosing a CPO with ISO 14001 certification and expertise in PCR materials, GreenRx ensured compliance while cutting costs. Small businesses can replicate this by auditing current medication packaging, requesting samples of PCR glass or compostable films, and conducting stability tests to verify drug safety. Promoting green credentials via a “Green Packaging” page or social media campaigns amplifies brand loyalty, cementing leadership in pharma packaging.
Practical Steps for Going Green
To embrace sustainable pharmaceutical packaging solutions, start with an audit of your current drug packaging to identify greener alternatives. Request samples of PCR glass or biodegradable plastics from your CPO and conduct rigorous stability tests to ensure compliance with FDA or EMA standards. Engage customers with transparent messaging, such as a dedicated “Green Packaging” page on your e-commerce site, detailing your commitment to sustainability. Partner with recycling programs like CiPPPA to align with PPWR mandates, and highlight these efforts in blog posts or Instagram campaigns to attract eco-conscious buyers. These steps position your brand as a 2025 leader in medication packaging, driving both sales and impact.
3-Smart Packaging: Embracing Technology
The Power of Digital Integration
Smart pharmaceutical packaging with RFID tags is revolutionizing the industry in 2025, blending technology with practicality to enhance traceability, combat counterfeiting, and engage patients. Systech’s UniSecure artAI, launched in February 2025, uses AI to verify packaging authenticity in real time, addressing the $200 billion counterfeit drug problem. RFID tags and IoT sensors enable real-time tracking of drug packaging, ensuring supply chain transparency, while NFC tags allow patients to access dosage instructions via smartphones. For small businesses, smart medication packaging offers a competitive edge, building trust and streamlining operations in a tech-driven market.
Opportunities for Small Businesses
For e-commerce pharmacies, smart pharmaceutical packaging with RFID tags unlocks new possibilities. IoT-enabled packaging monitors temperature during shipping, critical for biologics, while QR-coded labels reduce medication errors by linking to digital guides. In 2024, a small pharma brand partnered with Tjoapack to implement QR-coded labels, cutting errors by 15% and boosting customer trust. By outsourcing to CPOs with digital expertise, small businesses can adopt smart drug packaging without heavy R&D costs, positioning themselves as innovators. This is especially valuable for startups selling high-value drugs, where trust and precision are paramount.
PharmaEase’s Smart Packaging Triumph
PharmaEase, a startup specializing in migraine treatments, embraced smart medication packaging in 2024 by outsourcing to a CPO. NFC tags embedded in blister packs allowed patients to access dosage reminders via a mobile app, increasing adherence by 12% and repeat orders by 10%. By 2025, PharmaEase plans to integrate blockchain for supply chain transparency, a trend CPOs are increasingly offering. This case highlights how smart pharmaceutical packaging with RFID tags can transform patient outcomes and business growth. Small businesses can follow suit by identifying specific needs—serialization for compliance or IoT for monitoring—and partnering with tech-forward CPOs to implement scalable solutions.
Implementation Roadmap
To adopt smart drug packaging, identify your product’s requirements, such as serialization for anti-counterfeiting or IoT for cold chain monitoring. Partner with a CPO offering digital integrations like RFID or blockchain, and test prototypes to ensure user-friendliness. For example, IoT sensors in insulin vials can alert patients to temperature deviations, enhancing trust. By 2025, blockchain integration will provide end-to-end transparency, a boon for e-commerce pharmacies. Promote these features through targeted email campaigns or WordPress blog posts to highlight your brand’s innovation in pharmaceutical packaging, driving engagement and sales.
4- Cold Chain and Injectable Packaging: Powering Biologics
The Biologics Boom
The rise of biologics, accounting for 40% of the $1.5 trillion pharma market, is driving demand for cold chain pharmaceutical packaging for biologics in 2025. Biologics, including GLP-1 agonists for weight loss, require precise temperature control to maintain efficacy, making specialized medication packaging essential. Tjoapack’s 2024 expansion in cold chain and injectable services underscores the trend’s momentum, as CPOs invest in infrastructure to support this growing market. For small businesses, mastering cold chain drug packaging is critical to tapping into the $150 billion GLP-1 agonist market projected by 2030.
BioVita’s Cold Chain Success
BioVita, a startup e-commerce pharmacy, outsourced cold chain pharma packaging for a biologic drug in 2024, achieving remarkable results. The CPO provided temperature-controlled vials and real-time monitoring, reducing logistics costs by 15% and ensuring FDA compliance. This enabled BioVita to scale to 10,000 monthly orders, serving patients with chronic conditions like rheumatoid arthritis. By 2025, CPOs are expanding low-volume runs for personalized medicines, making cold chain pharmaceutical packaging for biologics accessible to niche players. BioVita’s success shows how small businesses can leverage CPO expertise to compete in high-value markets.
Strategic Execution for Small Businesses
To succeed with cold chain medication packaging, choose a CPO with proven expertise in biologics and cold chain logistics. Ensure scalable solutions to handle market surges, such as the projected $150 billion GLP-1 agonist demand. Request temperature stability data and compliance certifications, and integrate monitoring data into your e-commerce platform to build trust. For example, a dashboard displaying real-time temperature data reassures customers, boosting sales. Small businesses can differentiate by offering transparent, reliable drug packaging, positioning themselves as trusted providers in the biologics space.
Overcoming Logistics Challenges
Cold chain pharma packaging mitigates logistics risks like delays or temperature excursions, which can render biologics ineffective. Work with CPOs to develop contingency plans, such as backup shipping routes or emergency storage facilities, to ensure reliability. For e-commerce pharmacies, this reliability is a selling point, as patients demand consistent delivery of high-value drugs. By prioritizing robust cold chain drug packaging, small businesses can build a reputation for quality and trust, driving growth in 2025.
5- Personalized Medicine Packaging: Tailoring Solutions
The Pharmacogenomics Revolution
Personalized medicine, driven by pharmacogenomics (PGx), is reshaping medication packaging in 2025. As treatments become patient-specific, drug packaging must adapt to deliver variable dosing, tailored instructions, and low-volume runs for niche therapies. CPOs are leading the charge with custom blister packs, digitally printed labels for patient-specific information, and flexible production capabilities. This trend is particularly relevant for small businesses targeting high-value, personalized markets, where pharma packaging can enhance patient outcomes and brand loyalty.
SmartDose’s Personalized Breakthrough
SmartDose, a small biotech firm, partnered with a CPO in 2024 to develop custom blister packs for a PGx-based heart disease treatment. The drug packaging featured patient-specific dosing schedules and QR codes linking to personalized care plans, increasing adherence by 22%. By 2025, AI-driven tools are enabling hyper-customized medication packaging, such as blister packs optimized for elderly patients with color-coded dosing. SmartDose’s success highlights how small businesses can leverage personalized pharma packaging to create premium experiences that drive customer retention and market share.
Practical Steps for Implementation
To embrace personalized drug packaging, assess your product’s customization needs and partner with a CPO offering digital printing capabilities. Budget for low-volume runs to test market fit, and integrate with your e-commerce platform for seamless ordering. AI-designed blister packs with patient-specific features, like tactile indicators or large-print labels, enhance usability. Collect customer feedback to refine designs, ensuring they meet diverse needs. By promoting personalized medication packaging through targeted email campaigns or WordPress content, small businesses can foster loyalty and position themselves as innovators in pharmaceutical packaging.
Building Customer Loyalty
Personalized pharma packaging creates a premium experience that sets brands apart. For e-commerce pharmacies, this translates to higher retention and glowing reviews, as patients value tailored solutions that simplify their treatment. Highlight benefits like improved adherence in marketing campaigns, such as a blog post titled “How Personalized Drug Packaging Boosts Your Health Journey,” to maximize impact. By investing in personalized medication packaging, small businesses can build lasting connections with customers in 2025.
6-Nanotechnology Packaging: The Future Frontier
Emerging Innovations
Nanotechnology is an emerging frontier in drug packaging, offering precision for personalized therapies. In 2025, CPOs are experimenting with nano-coated vials that enhance drug stability and micro-packaging for targeted delivery. NanoRx, a small biotech firm, partnered with a CPO in 2024 to develop nano-coated syringes for a cancer therapy, improving drug efficacy by 10% and reducing waste. While still in early stages, nanotechnology holds immense potential for pharma packaging, particularly for high-value, low-volume drugs.
Opportunities for Startups
For startups, nanotechnology medication packaging offers a niche advantage in personalized medicine markets. Nano-coated vials or micro-packaging support small-batch production, ideal for e-commerce pharmacies targeting precision therapies. Partnering with CPOs investing in nanotechnology R&D allows small businesses to test prototypes without heavy investment, ensuring scalability and compliance. By adopting nanotechnology early, startups can position themselves as pioneers in drug packaging, gaining a competitive edge.
Scaling for the Future
Nanotechnology pharma packaging is poised for growth, with projections suggesting widespread adoption by 2030. Small businesses can pilot nano-coated vials for niche drugs, testing market fit with minimal risk. Collaborate with CPOs to ensure prototypes meet FDA or EMA standards, and promote these innovations through thought leadership content, such as a WordPress post on “Nanotechnology in Pharmaceutical Packaging: The Future Is Now.” Early adoption of nanotechnology drug packaging sets the stage for long-term success in a rapidly evolving industry.
Navigating Regulatory Compliance
Understanding the Regulatory Landscape
Pharmaceutical packaging compliance for small businesses in 2025 is shaped by complex regulations, including the EU’s FMD, FDA serialization mandates, and the PPWR. These rules aim to combat counterfeiting, ensure traceability, and promote sustainability, but pose significant challenges for startups with limited resources. CPOs provide expertise to navigate this landscape, reducing the risk of costly recalls and ensuring compliance with standards like ISO 22000 for food-grade packaging or ASTM D6400 for biodegradables. For small businesses, mastering drug packaging compliance is critical to building trust and avoiding penalties.
Practical Strategies for Compliance
To achieve compliance, partner with CPOs offering serialization solutions like RFID tags or blockchain for medication packaging. These technologies ensure traceability, meeting FMD and FDA requirements. Conduct regular audits to verify adherence to standards, and request compliance reports from your CPO to stay proactive. Monthly dashboards tracking serialization or material certifications help identify issues early. For example, embedding RFID tags in blister packs during production streamlines FMD compliance, saving time and costs. By leveraging CPO expertise, small businesses can turn pharma packaging compliance into a competitive advantage.
Anticipating Regulatory Shifts
Regulatory landscapes are evolving, with Asia-Pacific markets introducing stricter sustainability mandates by 2027. Engage with industry associations like the International Society for Pharmaceutical Engineering (ISPE) to stay ahead of changes. CPOs with strong industry ties provide early warnings, enabling small businesses to adapt drug packaging proactively. For instance, upcoming EU regulations may tighten recyclability standards, requiring shifts to PCR materials. By anticipating these changes, startups can avoid disruptions and maintain compliance in medication packaging.
VitaPack’s Compliance Victory
VitaPack, a small e-commerce pharmacy, avoided a costly recall in 2024 by partnering with a CPO that implemented blockchain-based serialization for its biologic drugs. The system ensured 100% traceability, meeting FDA requirements and boosting customer trust by 30%. This case underscores the value of pharmaceutical packaging compliance for small businesses, as compliance not only mitigates risks but also enhances brand reputation. Small businesses can replicate this by choosing CPOs with proven compliance track records and integrating serialization early in the drug packaging process.
Building a Compliance Framework
Create a tailored compliance checklist for your target markets (e.g., FDA for the US, EMA for Europe). Work with your CPO to embed serialization technologies, like RFID or QR codes, during packaging design. Regular training for your team on regulatory updates ensures long-term success. For example, a checklist covering FMD serialization and PPWR recyclability keeps your pharma packaging aligned with global standards, reducing risks and building trust with customers.
Selecting the Right CPO for Medication Packaging
-
Defining Your Packaging Needs
Choosing the right CPO is pivotal for effective drug packaging in 2025. Start by defining your product’s requirements—biologics, injectables, or oral drugs—and prioritize CPOs with specialized expertise. Verify compliance with FDA, EU, and ISO standards, and assess their capabilities in technologies like AI-driven serialization or IoT integration. For small businesses, a CPO that offers scalable solutions and low-volume runs is ideal, enabling flexibility as you grow. A thorough evaluation ensures your medication packaging aligns with your brand’s goals and market demands.
-
Evaluating Sustainability and Scalability
Sustainability is a non-negotiable in pharma packaging. Choose a CPO with ISO 14001 certification and expertise in sustainable pharmaceutical packaging solutions, such as PCR materials or biodegradable plastics. Scalability is equally critical, supporting low-volume runs for personalized medicines and high-volume projects for mass-market drugs. Request quotes, case studies, and virtual facility tours to gauge reliability. A CPO that balances sustainability and scalability ensures your drug packaging meets both consumer expectations and business needs.
-
RxPack’s CPO Success Story
RxPack, a small e-commerce pharmacy, partnered with a CPO in 2024 to develop sustainable medication packaging, reducing costs by 10% and earning a sustainability award. By shortlisting 3-5 CPOs and conducting virtual tours, RxPack ensured alignment with their goals. The CPO’s expertise in biodegradable blisters and low-volume runs allowed RxPack to launch a niche supplement line while maintaining compliance. Small businesses can follow this approach by researching CPOs through directories like Contract Pharma and prioritizing those with proven sustainability and scalability in pharma packaging.
-
Step-by-Step CPO Selection Process
-
Identify Needs: Determine your drug type (e.g., biologics, oral) and packaging requirements (e.g., cold chain, smart features).
-
Research CPOs: Use directories like Contract Pharma to shortlist 3-5 providers with relevant expertise.
-
Evaluate Capabilities: Request case studies, compliance certifications, and virtual facility tours.
-
Test Prototypes: Pilot packaging designs with real users to ensure functionality and appeal.
-
Negotiate Terms: Agree on KPIs like cost savings and defect rates to ensure long-term success.
-
Fostering Long-Term Partnerships
Establish clear communication channels, such as weekly check-ins, and define KPIs (e.g., cost savings, defect rates) to monitor CPO performance. A long-term partnership ensures consistent quality and innovation in drug packaging, critical for scaling your business. For example, regular feedback loops with your CPO can refine medication packaging designs, keeping them aligned with evolving consumer and regulatory demands.
Benefits of Optimizing Drug Packaging
1-Cost Efficiency and Compliance
Optimized medication packaging delivers 7-9% cost savings compared to in-house setups, freeing resources for small businesses to reinvest in growth. Compliance with regulations like FMD or FDA mandates reduces the risk of recalls, building trust with customers and regulators. By outsourcing to CPOs, startups achieve pharma packaging efficiency without the overhead of building facilities, making it a smart financial move.
2-Innovation and Market Agility
Access to innovations like AI, IoT, and biodegradable materials allows small businesses to compete without R&D investment. Scalable drug packaging supports market surges, such as the $150 billion GLP-1 agonist market by 2030. Faster market entry through CPO partnerships gives e-commerce pharmacies a competitive edge, enabling them to launch new products swiftly and capture market share.
4-Enhancing Brand Value
Sustainable pharmaceutical packaging solutions or smart pharmaceutical packaging with RFID tags differentiate brands, attracting eco-conscious or tech-savvy consumers. Highlight these benefits in marketing campaigns, such as a WordPress blog post titled “Why Our Drug Packaging Is Built for the Future,” to position your brand as a 2025 leader.
5-Driving Customer Trust and Retention
Effective medication packaging enhances trust through reliable delivery and user-friendly designs. A 2024 survey found that 65% of patients prefer brands with easy-to-use packaging, directly impacting repeat purchases for e-commerce pharmacies. By investing in pharma packaging that prioritizes user experience, small businesses can foster loyalty and turn one-time buyers into lifelong customers.
Overcoming Challenges in Medication Packaging
1-Balancing Sustainability and Safety
Achieving sustainability without compromising safety is a key challenge in drug packaging. Eco-friendly materials must meet stringent standards to protect drug efficacy. CPOs using ASTM D6400-certified biodegradable plastics or PCR glass mitigate risks through rigorous testing. Small businesses can partner with these CPOs to adopt sustainable pharmaceutical packaging solutions that comply with FDA or EMA standards, ensuring both safety and consumer appeal.
2- Managing Costs and Counterfeiting
Serialization costs for advanced medication packaging can strain small budgets, but CPOs offer scalable solutions to manage expenses. Counterfeit risks, costing the industry $200 billion annually, require robust technologies like Systech’s UniSecure artAI for authentication. By collaborating with CPOs, small businesses can implement cost-effective smart pharmaceutical packaging with RFID tags, safeguarding drugs and customers.
4- Mitigating Supply Chain Risks
Supply chain disruptions, such as material shortages, can derail pharma packaging. CPOs with diversified supplier networks, like multiple PCR glass providers, ensure continuity. For e-commerce pharmacies, this reliability prevents delays that could erode trust. By building resilient supply chains, small businesses can maintain consistent drug packaging quality, even in turbulent markets.
5-Addressing Scalability Challenges
Scaling medication packaging from niche to mass-market drugs is a hurdle for startups. CPOs offering flexible production runs—low-volume for personalized therapies, high-volume for generics—enable seamless growth. For example, a startup can begin with 1,000 custom blister packs and scale to 100,000 without retooling, keeping costs low. This flexibility makes pharma packaging a growth enabler for small businesses.
The Future of Pharmaceutical Packaging: 2030 and Beyond
-
Emerging Technologies
By 2030, drug packaging will be transformed by AI-driven design tools that optimize cost, sustainability, and user experience. Edible packaging, such as seaweed-based films, could replace blister packs, reducing waste by 40%. Nanotechnology will enable micro-packaging for hyper-personalized therapies, revolutionizing medication packaging for precision medicine. Small businesses that adopt these technologies early will gain a first-mover advantage in a $2 trillion market.
-
Regulatory and Global Trends
Global harmonization of serialization standards will simplify compliance, benefiting small businesses with pharma packaging. Circular recycling programs like CiPPPA will drive sustainability, with 60% of packaging expected to be recyclable by 2030. Asia-Pacific markets, projected to account for 35% of global drug packaging demand, will introduce stricter regulations, such as China’s Drug Administration Law. Early adopters of compliant, sustainable medication packaging will thrive in these high-growth regions.
-
Preparing for 2030
Partner with CPOs prioritizing R&D in pharma packaging, such as AI or nanotechnology. Attend conferences like Pharmapack Europe to stay ahead of trends like edible packaging or regulatory shifts. A forward-thinking strategy in 2025 ensures your business is ready for 2030’s innovations, positioning you as a leader in drug packaging.
-
FuturePharma’s Visionary Leap
FuturePharma, a startup, piloted edible packaging for oral supplements in 2025, reducing plastic waste by 50% and capturing 20% market share in eco-conscious segments. By partnering with a CPO specializing in sustainable innovations, FuturePharma stayed ahead of competitors. By 2030, edible and nano-based medication packaging are expected to dominate, making early adoption critical for small businesses in pharma packaging.
-
AI and Automation in Packaging
AI will streamline drug packaging by predicting demand, optimizing material use, and designing patient-specific solutions. For example, AI-driven tools can create blister packs tailored to a patient’s dosing schedule, reducing errors by 25%. Small businesses can access these technologies through CPO partnerships, avoiding in-house R&D costs. This democratization of innovation ensures startups remain competitive in medication packaging.
-
Tapping Global Markets
By 2030, Asia-Pacific’s 35% share of pharma packaging demand offers immense opportunities. Partner with CPOs with global networks to ensure compliance with regional regulations, such as India’s Drugs and Cosmetics Act. Early entry into these markets via scalable drug packaging positions small businesses for exponential growth, leveraging trends like biologics and generics.
Your Action Plan for Drug Packaging Success
To capitalize on pharmaceutical packaging in 2025, define your medication packaging needs—cold chain, sustainability, or smart tech. Research 3-5 CPOs using directories like Contract Pharma, and request quotes, case studies, and virtual tours. Test prototypes with target customers to ensure usability, and integrate drug packaging with your e-commerce platform for seamless ordering. Monitor metrics like cost savings (target 7-9%), customer satisfaction (aim for 20% increase), and adherence rates (goal: 15-20% improvement). For example, a pharmacy launching a biologic should prioritize CPOs with expertise in cold chain pharmaceutical packaging for biologics, testing prototypes to boost adherence and trust.
Implementation Timeline
-
Month 1: Audit current packaging, identify needs, and shortlist CPOs.
-
Month 2: Request prototypes and test with target customers.
-
Month 3: Finalize CPO partnership and integrate packaging into your e-commerce workflow.
-
Month 4: Launch marketing campaigns highlighting features like smart pharmaceutical packaging with RFID tags or sustainable pharmaceutical packaging solutions.
-
Ongoing: Monitor KPIs and refine packaging based on customer feedback.
Measuring Success
Use tools like Google Analytics to track traffic to your WordPress packaging pages, aiming for increased engagement and backlinks. Measure cost savings, customer satisfaction, and adherence improvements to quantify the impact of your pharma packaging strategy. For instance, a 15% adherence boost translates to higher retention and revenue for e-commerce pharmacies, proving the ROI of optimized drug packaging.
Frequently Asked Questions
What is pharmaceutical packaging?
Drug packaging involves materials and processes to protect, store, and deliver medications, ensuring safety, compliance, and user experience.
Why focus on medication packaging in 2025?
Pharma packaging saves 7-9% on costs, provides innovation, and ensures compliance in a $1.5 trillion market, ideal for small businesses.
How do I choose a CPO for drug packaging?
Assess expertise, sustainability, and scalability in medication packaging. Request case studies and virtual tours.
What trends are shaping pharmaceutical packaging in 2025?
Sustainable pharmaceutical packaging solutions, smart tech, cold chain, and personalized medicine drive the industry.
How can small businesses benefit from pharma packaging?
Optimized drug packaging offers cost savings, scalability, and innovation, enabling startups to compete.
What are the challenges of sustainable drug packaging?
Balancing safety and sustainability requires certified materials and rigorous testing in medication packaging.
Conclusion: Shape the Future with Drug Packaging
Pharmaceutical packaging in 2025 is a transformative opportunity for small businesses, e-commerce pharmacies, and industry professionals. With the market growing at a 7.1% CAGR, trends like sustainable pharmaceutical packaging solutions, smart pharmaceutical packaging with RFID tags, cold chain pharmaceutical packaging for biologics, and patient-centric pharmaceutical packaging for elderly are reshaping the industry. By optimizing drug packaging, you can save costs, stay compliant, and delight customers. Follow our action plan to find the right CPO and start today.